
|
Radioactivity in Nature

Radioactivity is a natural part of our environment. Present-day Earth contains all the stable chemical elements from the lowest mass (H) to the highest (Pb and Bi). Every element with higher Z than Bi is radioactive. The earth also contains several primordial long-lived radioisotopes that have survived to the present in significant amounts. 40K, with its 1.3 billion year half-life, has the lowest mass of these isotopes and beta decays to both 40Ar and 40Ca.
Many isotopes can decay by more than one method. For example, when actinium-226 (Z=89) decays, 83% of the rate is through b --decay, 226Ac Æ 226Th + e- + , 17% is through electron capture, 226Ac + e- Æ 226Fr + n , and the remainder, 0.006%, is through a-decay, 226Ac Æ 222Fr + 4He. Therefore from 100,000 atoms of actinium, one would measure on average 83,000 beta particles and 6 alpha particles (plus 100,000 neutrinos or antineutrinos). These proportions are known as branching ratios. The branching ratios are different for the different radioactive nuclei.
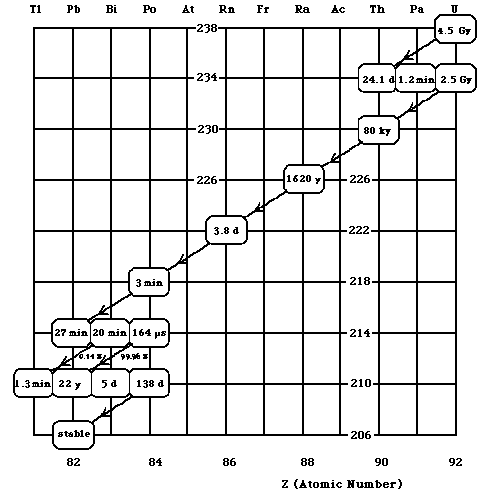
Three very massive elements, 232Th (14.1 billion year half-life), 235U (700 million year half-life), and 238U (4.5 billion year half-life) decay through complex "chains" of alpha and beta decays ending at the stable 208Pb, 207Pb, and 206Pb respectively. The decay chain for 238U is shown in Fig. 3-8. The ratio of uranium to lead present on Earth today gives us an estimate of its age (4.5 billion years). Given Earth’s age, any much shorter lived radioactive nuclei present at its birth have already decayed into stable elements. One of the intermediate products of the 238U decay chain, 222Rn (radon) with a half-life of 3.8 days, is responsible for higher levels of background radiation in many parts of the world. This is primarily because it is a gas and can easily seep out of the earth into unfinished basements and then into the house.
Some radioactive isotopes, for example 14C and 7Be, are produced continuously through reactions of cosmic rays (high energy charged particles from outside Earth) with molecules in the upper atmosphere. 14C is useful for radioactive dating (see Chapter 13). Also, the study of radioactivity is very important to understand the structure of the earth because radioactive decay heats the earth’s interior to very high temperatures.
|
|

